Why Pharmaceutical Waste Management Matters For Your Facility
Pharmaceutical waste includes any expired, unused, or leftover medications and materials that have come into contact with pharmaceuticals—from prescription bottles and IV bags to contaminated gloves and cleaning wastewater. Between 5 and 10 percent of all pharmaceutical waste qualifies as hazardous waste under EPA regulations, creating significant compliance obligations for facilities that generate it.
Quick Answer: What You Need To Know About Pharmaceutical Waste
- Definition: Discarded medications, chemical sludges, wastewaters from manufacturing, and contaminated items like syringes and PPE
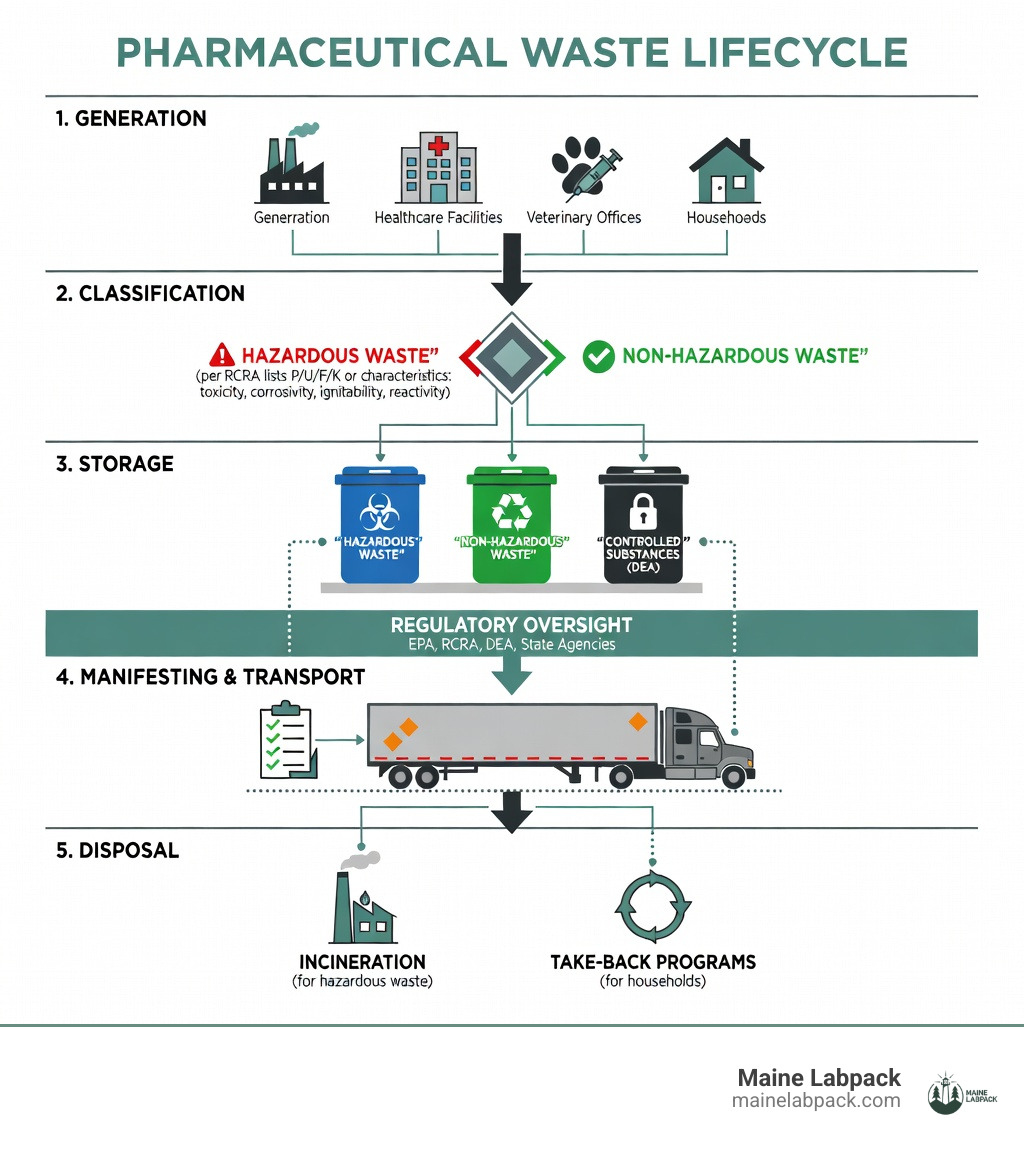
- Primary Generators: Healthcare facilities, pharmaceutical manufacturers, veterinary offices, and households
- Key Regulation: EPA’s Subpart P (2019) prohibits flushing hazardous pharmaceuticals and requires specific handling procedures
- Hazard Classification: 5-10% is hazardous based on EPA lists (P, U, F, K) or characteristics (toxicity, corrosivity, ignitability, reactivity)
- Disposal Methods: Professional waste services for facilities; take-back programs for households; never landfills or drains
If your medicine cabinet is full of expired drugs or your facility produces pharmaceutical waste, you’re facing both environmental and compliance challenges. Improper disposal releases active pharmaceutical ingredients into water systems and soil. It also creates risks for drug diversion—particularly with opioids and controlled substances.
The consequences of mismanagement are serious. Healthcare facilities that fail to properly classify and dispose of pharmaceutical waste face potential fines, regulatory violations, and operational disruptions. Even “non-hazardous” pharmaceutical waste requires careful handling since it can still harm human health and the environment, despite not being RCRA-regulated.
This guide walks you through everything you need to know: how to classify your pharmaceutical waste, which regulations apply to your facility, and what disposal methods keep you compliant while protecting your community.
Defining Pharmaceutical Waste And Its Primary Generators
To manage pharmaceutical waste effectively, you must first understand what it is and who generates it. In the simplest terms, it is any medicinal product that is no longer fit for its intended purpose. This isn’t just the pills in a bottle; it includes the sludges left over from making those pills, the water used to wash the vats, and the gloves worn by the nurse who administered the dose.
When these materials are handled improperly, the environmental risks are staggering. Active pharmaceutical ingredients (APIs) can leach into groundwater or pass through wastewater treatment plants that aren’t designed to filter out complex chemical compounds. This can lead to hormonally active water that affects aquatic life and, eventually, human health.
The primary generators of this waste include:
- Pharmaceutical Manufacturing Plants: These facilities produce large volumes of chemical sludges, spent solvents, and off-specification batches.
- Healthcare Institutions: Hospitals, clinics, and long-term care facilities deal with expired medications, partially used IV bags, and contaminated PPE.
- Veterinary Offices: Interestingly, veterinary offices often produce a more diverse array of pharmaceutical waste than human doctor’s offices because they perform a wider variety of procedures on-site, from surgeries to respiratory treatments.
- Households: Every medicine cabinet in New England is a potential source of waste.
For facilities handling highly regulated substances, managing Controlled DEA Waste adds another layer of complexity, requiring strict security to prevent drug diversion and abuse.
Identifying Different Types Of Pharmaceutical Waste
Not all waste is created equal. To stay compliant, generators must distinguish between various physical and chemical forms of waste.
- Solid Waste: This includes pills, capsules, tablets, and even sharps like needles that might have drug residue on them.
- Liquid Waste: Syrups, intravenous fluids, and the liquid medications found in vials.
- Chemical Sludges And Wastewater: Primarily a concern for manufacturers, these are the byproducts of the chemical synthesis process.
- Contaminated PPE And Items: Rags, mop heads, gloves, and gowns that have come into contact with hazardous drugs.
According to the Environment Agency guidance on defining pharmaceutical waste, it also includes any medicine that is out of date, damaged, or no longer required. Even empty containers can be considered waste if they haven’t been managed according to specific “empty” standards.
The Regulatory Landscape Of Pharmaceutical Waste
Navigating the rules for pharmaceutical waste can feel like trying to read a prescription written in shorthand. The primary authority in the United States is the Environmental Protection Agency (EPA), which uses the Resource Conservation and Recovery Act (RCRA) to regulate hazardous waste from “cradle to grave.” This means the moment a waste is generated, the facility is responsible for it until it is safely destroyed.
In 2019, the EPA finalized a landmark set of regulations known as Subpart P. This rule was designed specifically for healthcare facilities and reverse distributors, recognizing that the old industrial hazardous waste rules didn’t quite fit the environment of a hospital or pharmacy.
Knowing How to Securely Destruct Pharmaceutical Waste is now a legal requirement, not just a suggestion.
Managing Hazardous Pharmaceutical Waste Under Subpart P
Subpart P introduced several major changes that healthcare facilities in Maine, New Hampshire, and across New England must follow. One of the most significant is the sewering ban. It is now strictly prohibited to flush hazardous waste pharmaceuticals down the drain. While the ban technically applies to hazardous waste, the EPA and state agencies strongly discourage sewering any pharmaceuticals due to the environmental impact.
Subpart P also changed how facilities are categorized. Under the old rules, a single P-listed waste (like a nicotine patch) could bump a small doctor’s office into a Large Quantity Generator status, bringing a mountain of paperwork with it. Now, hazardous waste pharmaceuticals managed under Subpart P do not count toward your monthly generator category determination. This is a huge relief for many facilities, though it requires opting into the Subpart P standards.
For those unsure of how to start, the EPA provides a Step-by-Step Guide to Notifying Under Subpart P to help facilities properly register their activities.
Overlap Between RCRA And DEA Regulations
Things get even stickier when a drug is both an RCRA hazardous waste and a DEA controlled substance. Think of drugs like phenobarbital or certain testosterone gels. These are dual waste streams.
To stay compliant, you must satisfy both agencies. This typically means:
- Storing the waste in a secure, DEA-compliant manner.
- Using a disposal method that meets the DEA “non-retrievable” standard (often through specialized incineration).
- Maintaining records that satisfy both RCRA tracking and DEA inventory requirements.
Conducting Hazardous Waste Due Diligence Healthcare is essential to ensure your facility isn’t accidentally violating one set of rules while trying to follow another.
Classifying Hazardous vs. Non-Hazardous Materials
How do you know if that bottle of pills is “hazardous” or just “waste”? The EPA uses two main methods for classification: Lists and Characteristics.
Listed Wastes:
- P and U lists: These are commercial chemical products. The P-list contains “acute” hazardous wastes (very dangerous even in small amounts), while the U-list contains “non-acute” hazardous wastes. Examples include Epinephrine (P042) and certain chemotherapy drugs, such as Cyclophosphamide (U058).
- F and K lists: These generally apply to manufacturing processes rather than finished drugs.
Characteristic Wastes (The “D” List): If a drug isn’t on a list, it might still be hazardous if it exhibits one of these four traits:
| Characteristic | Definition | Pharmaceutical Example |
|---|---|---|
| Ignitability | Can catch fire easily (flash point <140°F). | Alcohol-based cough syrups, aerosols. |
| Corrosivity | Can burn skin or corrode metal (pH ≤2 or ≥12.5). | Certain compounding acids or bases. |
| Reactivity | Unstable, reacts violently with water/air. | Nitroglycerin (in certain forms). |
| Toxicity | Harmful if swallowed or absorbed (fails TCLP test). | Meds containing Mercury, Selenium, or Silver. |
You can find more details on these categories in the P and U lists documentation.
Best Practices For Healthcare Facilities And Reverse Distributors
Compliance isn’t just about knowing the rules; it’s about what you do on the floor every day. Most healthcare facilities produce only 5% to 15% hazardous waste, but that small percentage requires 100% of your attention.
Generator Categories: Facilities are classified as Very Small (VSQG), Small (SQG), or Large Quantity Generators (LQG). Under Subpart P, healthcare facilities have a more streamlined path, but they must still notify the EPA of their status.
Color-Coded Containers: A simple way to prevent mistakes is to use standardized containers:
- Black Containers: Hazardous waste pharmaceuticals.
- Blue Containers: Non-hazardous pharmaceutical waste.
- Yellow Containers: Trace chemotherapy waste.
- Red Containers: Biohazardous/Sharps waste.
Manifesting and Shipping: When shipping non-creditable hazardous waste off-site, you must use a Uniform Hazardous Waste Manifest. A unique requirement for this waste is the use of the PHRM code in the manifest’s fields, which helps waste facilities identify and handle the material correctly. More information can be found on Manifesting Non-Creditable Hazardous Waste Pharmaceuticals.
Creditable vs. Non-Creditable:
- Potentially Creditable: Unused meds in original packaging that might be eligible for manufacturer credit. These are sent to a reverse distributor.
- Non-Creditable: Broken, expired, or used meds that must be sent directly to a disposal facility.
Household Disposal And Community Take-Back Programs
For the average person in Manchester, NH, or Burlington, VT, the rules are much simpler, but the stakes are just as high. Keeping unused meds—especially opioids—in the house increases the risk of accidental poisoning and addiction.
The gold standard for household disposal is a Take-Back Program. The DEA sponsors National Prescription Drug Take Back Day twice a year, providing safe, anonymous drop-off locations. Many local police departments and pharmacies also have permanent drop-off kiosks.
If a take-back program isn’t available, follow these steps for the trash:
- Mix the meds with something unappealing, like used coffee grounds or kitty litter.
- Place the mixture in a sealed bag or container.
- Scratch out your personal information on the original prescription bottle.
- Throw it in the household trash.
The “Flush List”: While sewering is generally a “no-no,” the FDA maintains a very small “flush list” for medications that are so dangerous they must be destroyed immediately if a take-back option isn’t available (like Fentanyl patches). The FDA published a paper finding that the environmental risk from flushing these specific drugs is negligible compared to the risks of theft or accidental ingestion.
For those looking for a simple home solution, you can Request a Free Sample of Rx Destroyer, which renders medications unusable and safe for disposal.
Frequently Asked Questions About Pharmaceutical Waste
How should empty pharmaceutical containers and syringes be managed?
Managing “empty” containers is a common point of confusion. Under the new rules, a container that held a hazardous pharmaceutical is considered “RCRA empty” (and therefore non-hazardous) if:
- Stock bottles/vials: The contents have been removed using standard methods (e.g., pouring or pumping), and no more than 3% of the capacity remains.
- Syringes: The plunger has been fully depressed. These should still be placed in a sharps container for safety.
- IV Bags: The liquid has been fully administered to the patient.
However, if the container held “P-listed” waste, the rules were much stricter (requiring triple rinsing). Subpart P has simplified this, but it is vital to check the specific guidance on Containers that Once Held P-listed Pharmaceutical Waste to ensure you aren’t over-classifying your trash.
What is the status of nicotine replacement therapies under the P075 listing?
For years, nicotine was a major headache for hospitals because it was listed as an acute hazardous waste (P075). This meant every discarded nicotine patch or piece of gum had to be handled like toxic sludge.
The 2019 rule provided a much-needed exemption: FDA-approved over-the-counter (OTC) nicotine replacement therapies (NRTs)—like patches, gums, and lozenges—are no longer considered P075 hazardous waste.
Note: This exemption does not apply to e-liquids, vape pens, or prescription nicotine. Those must still be managed as hazardous waste. However, Nicotine-containing products that are legitimately recycled are not considered solid waste, which opens up more sustainable disposal options for those specific items.
Why is sewering hazardous waste pharmaceuticals prohibited?
The ban on flushing drugs and the water supply exists because our infrastructure wasn’t built for modern chemistry. Most municipal wastewater treatment plants use biological processes—bacteria eating organic matter—to clean water. They are not equipped to break down complex molecules like antibiotics, hormones, or chemotherapy agents.
When these drugs pass through the system, they enter our rivers and lakes. This can lead to antibiotic resistance in environmental bacteria and “feminization” of fish populations due to endocrine disruptors. By keeping these chemicals out of the drain, we protect the New England water table and ensure our local ecosystems remain healthy.
Turning Compliance Into Safer, Simpler Disposal
Managing pharmaceutical waste is more than just a regulatory hurdle; it’s a commitment to the health of your staff, your patients, and the New England environment. From the bustling hospitals of Boston and Worcester to the quiet veterinary clinics of Northern Maine and the Green Mountains of Vermont, every generator plays a role in keeping hazardous chemicals out of our water and air.
The rules are complex, but you don’t have to navigate them alone. Whether you are a Large Quantity Generator trying to implement Subpart P or a small clinic looking for a more sustainable way to handle your expired meds, professional help is available.
Maine Labpack specializes in providing professional, on-time, and sustainable hazardous waste management solutions across Maine, New Hampshire, Massachusetts, Rhode Island, and Vermont. As a one-stop shop that comes directly to your site, they ensure your facility remains in total compliance with EPA, DEA, and state regulations while minimizing your environmental footprint.
Ready to simplify your disposal process? Manage your pharmaceutical waste with Maine Labpack and let the experts handle the chemistry so you can focus on your patients.